1. Acyloin Ester Condensation
Alpha-Hydroxyketones from carboxylic esters
Upon heating of a carboxylic ester with sodium in an inert solvent, a conden-sation reaction can take place to yield a alpha-hydroxy ketone after hydrolytic workup.
This reaction is called Acyloin condensation, named after the products thus obtained. It works well with alkanoic acid esters. For the synthesis of the corresponding products with aryl substituents (R=aryl),the Benzoin condensation of aromatic aldehydes is usually applied.
For the mechanistic course of the reaction the diketone 5 is assumed to be an intermediate, since small amounts of 5 can sometimes be isolated as a minor product. It is likely that the sodium initially reacts with the ester 1 to give the radical anion species 3, which can dimerize to the dianion 4. By release of two alkoxides RO Negative the diketone 5 is formed. Further reaction with sodium leads to the dianion 6, which yields the alpha-hydroxy ketone 2 upon aqueous workup.
An intramolecular reaction is possible with appropriate substrates containing two ester groups, leading to the formation of a carbocyclic ring. This reaction is especially useful for the formation of rings with ten to twenty carbon atoms, the yield depending on ring size. The presence of carbon–carbon double or triple bonds does not affect the reaction. The strong tendency for ring formation with appropriate diesters is assumed to arise from attachment of the chain ends to the sodium surface and thereby favoring ring closure.
A modified procedure, which uses trimethylsilyl chloride as an additional reagent, gives higher yields of acyloins and is named after Ruhlmann. In the presence of trimethylsilyl chloride, the bis-O-silylated endiol 7 is formed and can be isolated.
Treatment of 7 with aqueous acid leads to the corresponding acyloin.
2. Baeyer–Villiger Oxidation
Oxidation of ketones to carboxylic esters. When a ketone 1 is treated with hydrogen peroxyde or a peracid, a formal insertion of oxygen can take place to yield a carboxylic ester 2. This process is called the Baeyer–Villiger oxidation.
In a first step the reactivity of the carbonyl group is increased by protonation at the carbonyl oxygen. The peracid then adds to the cationic species 3 leading to the so-called Criegee intermediate.
Cleavage of the carboxylic acid R3COOH from that intermediate leads to an electron-deficient oxygen substituent with an electron sextet configuration. This deficiency can be compensated through migration of the substituent R1 or R2; experimental findings suggest that cleavage and migration are a concerted process. The cationic species 5 which can be thus formed (e.g. by migration of R2), loses a proton to yield the stable carboxylic ester 2:
The ease of migration of substituents R1, R2 depends on their ability to stabilize a positive charge in the transition state. An approximate order of migration has been drawn: R3C > R2CH > Ar > RCH2 > CH3. Thus the Baeyer–Villiger oxidation of unsymmetrical ketones is regioselective. On the other hand aldehydes usually react with migration of the hydrogen to yield the carboxylic acid.
The reaction mechanism is supported by findings from experiments with 18-O labeled benzophenone 6; after rearrangement, the labeled oxygen is found in the carbonyl group only:
The Dakin reaction proceeds by a mechanism analogous to that of the baeyer–Villiger reaction. An aromatic aldehyde or ketone that is activated by a hydroxy group in the ortho or para position, e.g. salicylic aldehyde 12 (2-hydroxybenzaldehyde), reacts with hydroperoxides or alkaline hydrogen peroxide. Upon hydrolysis of the rearrangement product 13 a dihydroxybenzene, e.g. catechol 14, is obtained. The electron-donating hydroxy substituent is necessary in order to facilitate the migration of the aryl group; otherwise a substituted benzoic acid would be obtained as reaction product.
3. Beckmann Rearrangement
Rearrangement of oximes to give N-substituted carboxylic amides. The rearrangement of oximes 1 under the influence of acidic reagents to yield N-substituted carboxylic amides 2, is called the Beckmann rearrangement. 1,2 The Reaction is usually applied to ketoximes, aldoximes often are less reactive.
Upon treatment with a protic acid, the hydroxy group of the oxime 1 initially is protonated to give an oxonium derivative 3 which can easily lose a water molecule. The migration of the substituent R (together with the bonding electrons) and loss of water proceed simultaneously.The cationic species 4 thus formed reacts with water to give the iminol 5, which tautomerizes to a more stable amide tautomer, the N-substituted carboxylic amide-2. Those steps correspond to the formation of amides by the Schmidt reaction.
A side reaction can give rise to the formation of nitriles.
As reagents concentrated sulfuric acid, hydrochloric acid, liquid sulfur dioxide,
thionyl chloride, phosphorus pentachloride, zinc oxide4 and even silica gel-5 can be used. Reagents like phosphorus pentachloride (as well as thionyl chloride and others) first convert the hydroxy group of the oxime 1 into a good leaving group.
The stereochemical course of the Beckmann rearrangement often allows for the prediction of the reaction product to be obtained; in general the substituent R anti to either the hydroxy or the leaving group will migrate:
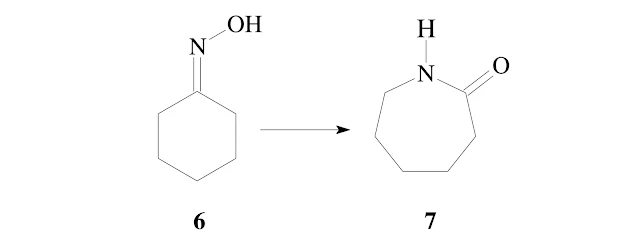
In some cases a mixture of the two possible amides may be obtained. This has been rationalized to be a result of partial isomerization of the oxime under the reaction conditions, prior to rearrangement. With aldoximes (R D H) a migration of hydrogen is seldom found. The Beck-mann rearrangement therefore does not give access to N-unsubstituted amides. The reaction with oximes of cyclic ketones leads to formation of lactams (e.g.6>7) by ring enlargement.
This particular reaction is performed on an industrial scale; ε-caprolactam 7 is used as monomer for polymerization to a polyamide for the production of synthetic fibers.
Substituents R, R' at the starting oxime 1 can be H, alkyl, or aryl. The reaction conditions for the Beckmann rearrangement often are quite drastic (e.g.concentrated sulfuric acid at 120 °C), which generally limits the scope to less sensitive substrates. The required oxime can be easily prepared from the respective aldehyde or ketone and hydroxylamine.
4. Benzidine Rearrangement
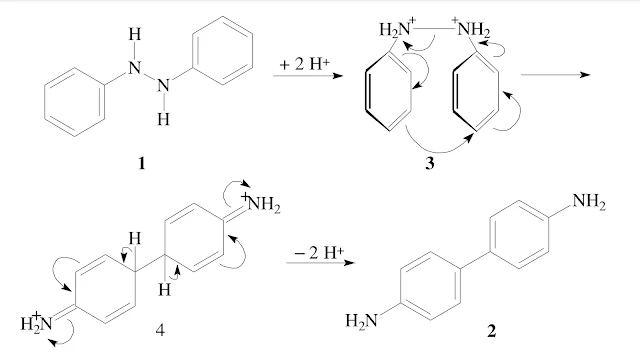
Rearrangement of hydrazobenzene to yield benzidine. Hydrazobenzene 1 (1,2-diphenyl hydrazine) is converted to benzidine 2 (4,4'-diaminobiphenyl) under acidic conditions. This unusual reaction is called the benzidine rearrangement,and can be observed with substituted diphenyl hydra-zines as well.
In accord with the experimental findings a mechanism via a [5,5]-sigmatropic rearrangement has been formulated. In a first step the hydrazobenzene is protonated to the dicationic species 3, in which the phenyl groups can arrange in such a way to allow for rearrangement:
The reaction can be first or second order with respect to the H+ concentration.
In weakly acidic solution it is first order in [H+], but second order in strongly acidic solution. This indicates that the monoprotonated as well as the diprotonated hydrazobenzene can undergo rearrangement.
The rearranged dicationic species 4, which has been shown to be an intermediate-7, leads to the stable benzidine 2 upon deprotonation. It has been demonstrated by crossover experiments that the rearrangement does not proceed via a dissociation/recombination process. From the reaction of hydrazobenzene the benzidine is obtained as the major product (up to 70% yield), together with products froms Side reactions—2,4'-diaminobiphenyl 5 (up to 30% yield) and small amounts of 2,2'-diaminobiphenyl 6 as well as o- and p-semidine 7 and 8:
The rearrangement takes place in the presence of strong mineral acids (e.g.hydrochloric or sulfuric acid) in aqueous solution or water–alcohol mixtures at room temperature; in some cases slight warming may be necessary. The benzidine rearrangement is of interest for mechanistic considerations.The preparative applicability may be limited because of the many side products,together with low yields. Furthermore benzidine is a carcinogenic compound.























0 Comments